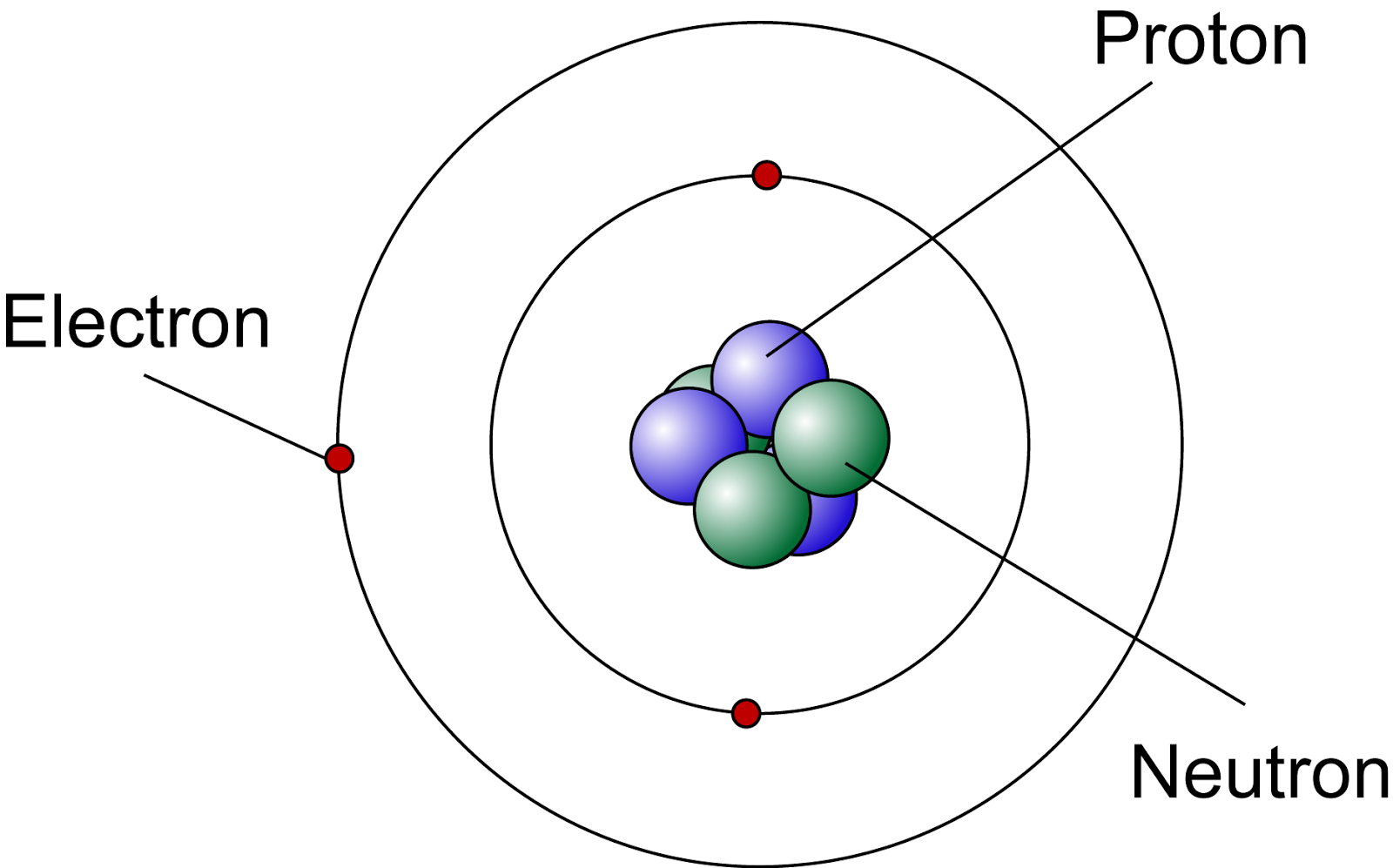
The charge of an atom is defined as follows:Ītomic charge = number of protons − number of electronsĪs will be discussed in more detail later in this chapter, atoms (and molecules) typically acquire charge by gaining or losing electrons. When the numbers of these subatomic particles are not equal, the atom is electrically charged and is called an ion. (This isotope is known as “carbon-12” as will be discussed later in this module.) Thus, one amu is exactly \fracĪtoms are electrically neutral if they contain the same number of positively charged protons and negatively charged electrons. Since 1961, it has been defined with regard to the most abundant isotope of carbon, atoms of which are assigned masses of exactly 12 amu. The amu was originally defined based on hydrogen, the lightest element, then later in terms of oxygen. When describing the properties of tiny objects such as atoms, we use appropriately small units of measure, such as the atomic mass unit (amu) and the fundamental unit of charge (e). For example, a carbon atom weighs less than 2 × 10 −23 g, and an electron has a charge of less than 2 × 10 −19 C (coulomb). (credit middle: modification of work by “babyknight”/Wikimedia Commons credit right: modification of work by Paxson Woelber)Ītoms-and the protons, neutrons, and electrons that compose them-are extremely small. If an atom could be expanded to the size of a football stadium, the nucleus would be the size of a single blueberry. For a perspective about their relative sizes, consider this: If the nucleus were the size of a blueberry, the atom would be about the size of a football stadium ( Figure 1). The diameter of an atom is on the order of 10 −10 m, whereas the diameter of the nucleus is roughly 10 −15 m-about 100,000 times smaller. The nucleus contains the majority of an atom’s mass because protons and neutrons are much heavier than electrons, whereas electrons occupy almost all of an atom’s volume. It was learned that an atom contains a very small nucleus composed of positively charged protons and uncharged neutrons, surrounded by a much larger volume of space containing negatively charged electrons. The development of modern atomic theory revealed much about the inner structure of atoms. Calculate average atomic mass and isotopic abundance.Define the atomic mass unit and average atomic mass.Write and interpret symbols that depict the atomic number, mass number, and charge of an atom or ion.The answer is that the electric field is coming out of this fear.By the end of this section, you will be able to: Also normal to the sphere radiantly So in question, be since the proteins air positive. And if we have a negative charge, then the electric field will be coming in. We also have to calculate the direction, notice that if we have a positive charge then the electric fields will be coming out of the charge radial e normal to the surface of this year. I understand to the 21 Newton's recall, Um, and this is the answer to question A. So the electric feud in magnitude it's equal to three point of seven. So that's 6.64 times 10 to the minus 15 m squared. Then we have to divide it by the radius squared off this year. That's 94 proteins, which broaden has charged at one point six times 10 to the minus 19 columns. No que is 9.9 times scents of the ninth Newtons meters squared, curriculum squared times the charge. So the mhm in the electric field is Q over four pi R squared Absence your now 1/4 pi up zero a school ums constant K That's nine times 10 to the nine um, Newton's There's your Newtons meters squared Curriculum Square so the electric field is k que over our square. 
So that's e times four pi r squared, and this is equal to the total charge over absolute zero. Notice that if the atom is a sphere than we have a spherical symmetry, meaning that the integral off E. And given this, our goal is to calculate what is the electric field just outside the surface off the nucleus? Yeah, s o, I don't if I said before that are was the readers of the nucleus of the atom. Uh, the atom is plutonium that has 94 proteins. So we have thio approximate the atom biosphere with a radius of zero of 6.64 times, 10 to the minus 15 m. Okay, eso in our problem, we're going to calculate the electric field generated by an atom just at its surface. We're absolutely zero is the vacuum permeability. The integral of the electric field dot the service element DS over all the surface physical to Q over absolute zero. What God's law tells us is that the electric flux over the surface So we define that as the electric field. And we draw a caution surface around discharge distribution. We're going to talk about houses to consider that we have a charge distribution that has a total charge.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
